First targeted treatment for low-grade brain tumors on the horizon

Results from a multi-institutional phase III trial indicate an increase in progression-free survival
This summer at the American Society of Clinical Oncology Annual Meeting, scientists presented results from a phase III clinical trial showing that a drug called vorasidenib slowed the growth of a subtype of grade 2 diffuse gliomas. With these findings, which were simultaneously published in the New England Journal of Medicine, the drug’s manufacturer Servier is now seeking regulatory approval from the Food and Drug Administration.
Here’s what you need to know about these results and what they could mean for patients with low-grade brain tumors.
What makes low-grade gliomas difficult to treat?
Unlike in high-grade gliomas, most people with lower grade brain tumors tend to be under 50 years old when they are diagnosed.
“These tend to be younger people – people who are planning their families, early in their careers, or figuring out school,” said UCSF neuro-oncologist Jennie Taylor, MD, MPH “so you're trying to strike the balance of preserving quality of life with knowing that you’re going to need some of these intensive treatments – like maybe another surgery or radiation or chemotherapy – at some point.”
While low-grade gliomas are generally slow growing, these tumors can also progress very quickly in some patients despite what physicians would predict from their biopsy sample alone, she said.
Mutations in the isocitrate dehydrogenase (IDH) 1 and 2 genes are now key criteria in the 2021 World Health Organization’s classification scheme for different glioma subtypes.
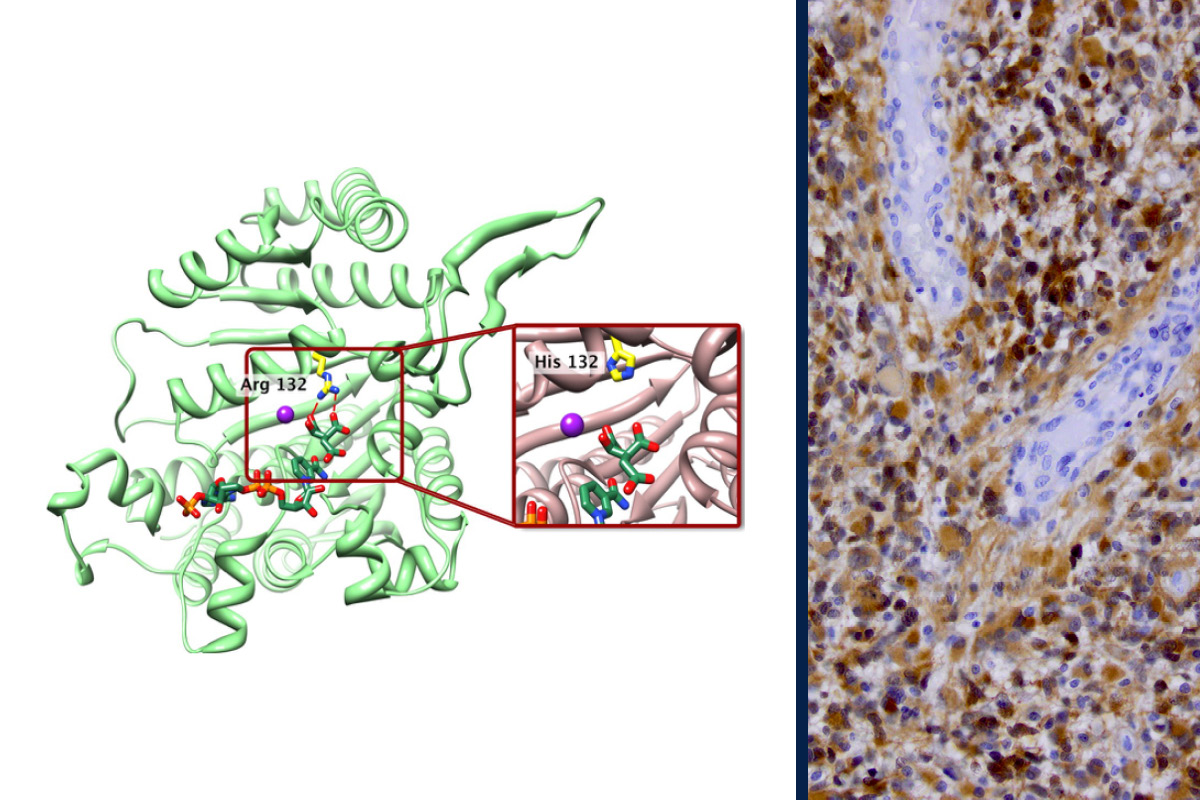
“In 2009 – when we first found this IDH mutation – it really helped clarify who those different people were going to be, and we could figure that out at the time of their original surgery,” Taylor said.
How does this drug work?
Discovering these IDH mutations helped scientists identify new therapeutic strategies to target brain tumors since the mutations are present in other, more prevalent types of cancers.
Oncologists already use an inhibitor of the mutant IDH1, to treat certain types of leukemias and cholangiocarcinomas. Researchers compared this drug, ivosidenib, to vorasidenib, which inhibits both the IDH1 and IDH2 mutations and is designed for better uptake across the blood-brain barrier into brain tumors. The findings from that phase I clinical trial, published earlier this year in Nature Medicine, showed that the tumors from patients taking vorasidenib got into the brain and decreased production of 2-hydroxyglutarate (2-HG), the byproduct of the IDH mutations that scientists think helps drive tumor growth. Encouraging early results also showed that some patients responded to treatment with tumor shrinkage.
“That peri-operative study really gave us a clear signal of how impactful this treatment might be,” Taylor said.
What are the key findings from the trial?
The clinical trial recruited 331 people with grade 2, IDH1/2-mutant gliomas from January 2020 to February 2022 at multiple study sites, including the UCSF Brain Tumor Center. Patients were randomized to either vorasidenib or placebo and followed every three months with MRIs. The trial participants had only received surgery. They were in what oncologists call a “watchful waiting” period, not receiving any other type of brain tumor therapy.
Based on analyses of their MRI scans, people who received vorasidenib went longer before experiencing disease progression compared to those who received the placebo control – 27.7 months versus 11.1 months. Individuals who received the drug also had more time before needing new treatment – such as crossing over from placebo to vorasidenib, another surgery, radiation, or chemotherapy.
“We’re all cautiously optimistic, but early on it seems like vorasidenib certainly helped slow down tumor growth,” Taylor said.
The drug, which patients take as a daily pill, was also well-tolerated with few side effects that interfered with an individual’s quality of life.
"Compared to chemotherapy – for example, temozolomide or CCNU – there’s not a lot of nausea, increased risk of infection, bleeding, or all the things you typically worry about,” she said.
What could this drug mean for patients with low-grade brain tumors?
Taylor stresses that vorasidenib is not a cure for the disease. Although these initial results suggest the drug is slowing brain tumor growth, it will be years before scientists know how the drug affects survival outcomes. Researchers are also still in the process of collecting data on other outcomes – like if it improves cognition or seizure control.
Brain tumor researchers also don’t know how long people need to be on this medication, which presents its own challenge in these younger patients.
“The timing of and the consequences of putting particularly young patients on this drug, which may interfere with family planning, is something that we’re going to have to be really mindful of,” Taylor said.
But she thinks that the drug may help mitigate many challenges in treating low-grade gliomas.
“Because living with the disease is such a marathon, we’re really trying to place the water stations – things like radiation, another surgery, and chemotherapy – strategically,” she said. “The hope about vorasidenib is that it allows us to even further delay some of these more intensive treatments that we worry can negatively impact quality of life.”
The rigorous, standardized process that the scientists are using to interpret the data from thousands of MRI scans across multiple institutions over many years, Taylor says, could also advance how physicians monitor disease progression.
What about other IDH-mutant gliomas?
“The population that they set out to investigate in this trial is still very specific. It's patients who have grade 2 tumors, and who are one to five years out from surgery,” Taylor said. “But there are a lot more people living with IDH-mutant tumors who don't fall into that category.”
Currently, scientists do not yet know who else may benefit from taking vorasidenib, but patients may have the opportunity to receive the drug before it is approved by the FDA through an expanded access program.
Additionally, new clinical studies are underway targeting other patient populations. For example, a new phase I clinical trial is now recruiting patients at UCSF with both grade 2 and 3 IDH-mutant recurrent astrocytomas to study the safety and preliminary efficacy of vorasidenib when used in combination with an immunotherapeutic drug called pembrolizumab around the time of surgery for disease recurrence. By analyzing the patients’ tumor samples, the researchers will get early indication of how the tumor is responding to the combination of therapies.
Taylor says she is also interested in seeing how vorasidenib might be used in combination with radiation and chemotherapies to perhaps improve survival outcomes for patients.
“In my decade career as a neuro-oncologist, this is the most exciting thing I’ve ever seen,” she said. “I think it will open up a whole new world of how to target this really key IDH mutation and help our patients hopefully live longer and better.”