New UCSF study develops a brain tumor organoid model to test potential treatment approaches
The many cell types within meningiomas don’t all necessarily respond to therapies in the same way, which complicates current strategies to treat these brain tumors. Scientists need a more accurate representation of how all these cells, with their unique genetic and biological properties, interact.
Researchers at UC San Francisco have now identified mechanisms high-grade meningiomas use to evade treatments. The findings, published on May 17 in Nature Genetics, helped them to develop a cell culture model for studying the way these tumors grow and for screening different drugs in the lab.
“The tumor cells all contribute something different — with some becoming more important than others as the tumor grows and responds to treatment,” said UCSF physician-scientist and the study’s senior author David Raleigh, MD, PhD. “We need to identify those reservoirs of recurrence and target those cells to alter the trajectory of a tumor’s lifespan.”
Putting genetic mutations in context
The World Health Organization classifies high-grade meningiomas based on what they look like under the microscope as well as the presence of a handful of genetic mutations that drive tumor formation.
Raleigh and his colleagues suspected that grouping tumors with these mutations together might be masking important distinctions in their biology and, by extension, potential therapeutic vulnerabilities.
Conventional techniques for studying how genetic mutations in tumors go on to affect gene expression and protein levels in cells involve processing tissue specimens in a test tube. These approaches offer aggregate measures of the abundance of all the genes and proteins present in the tumor but are divorced from the specifics of which cells have those molecular features.
“We're very good at tracking the DNA mutations in tumors, but the biochemical and cellular consequences of those mutations are often complex and very context dependent,” Raleigh, who is also the Robert and Ruth Halperin Endowed Chair in Meningioma Research, said.
To better understand the impact of the different mutations driving tumor growth, the scientists — led by former UCSF neuro-pathology fellow Calixto-Hope Lucas, MD — turned to new technology that enabled them to visualize differences in gene expression across 16 tissues samples from 10 meningiomas.
With the help of collaborators at Northwestern University, Raleigh and his team were also able to see how those alterations in the genomic and transcriptomic architecture of the tumor cells then influence protein levels and protein signaling events.
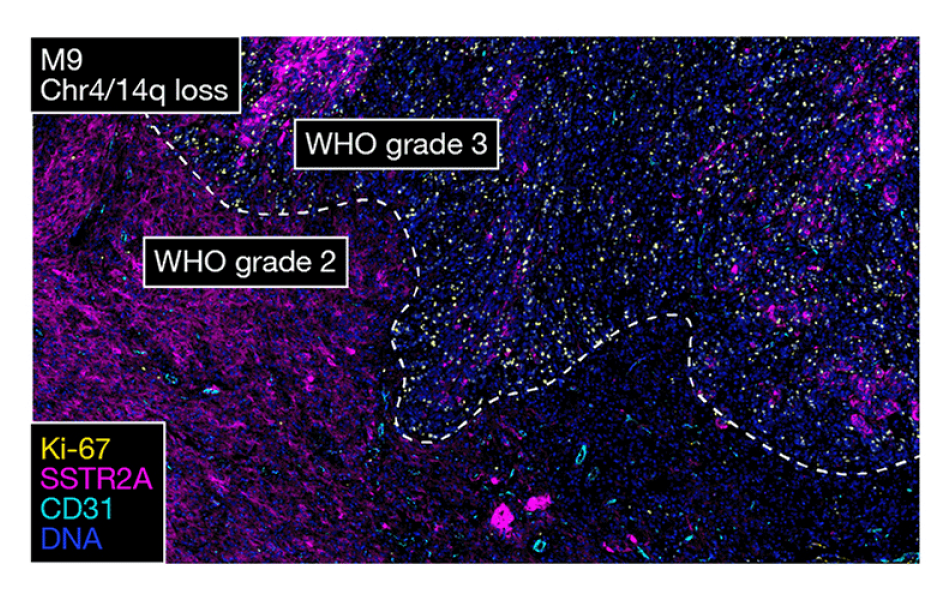
High-grade meningiomas have distinct gene and protein expression patterns compared to lower grade meningiomas. Image courtesy of the Raleigh lab.
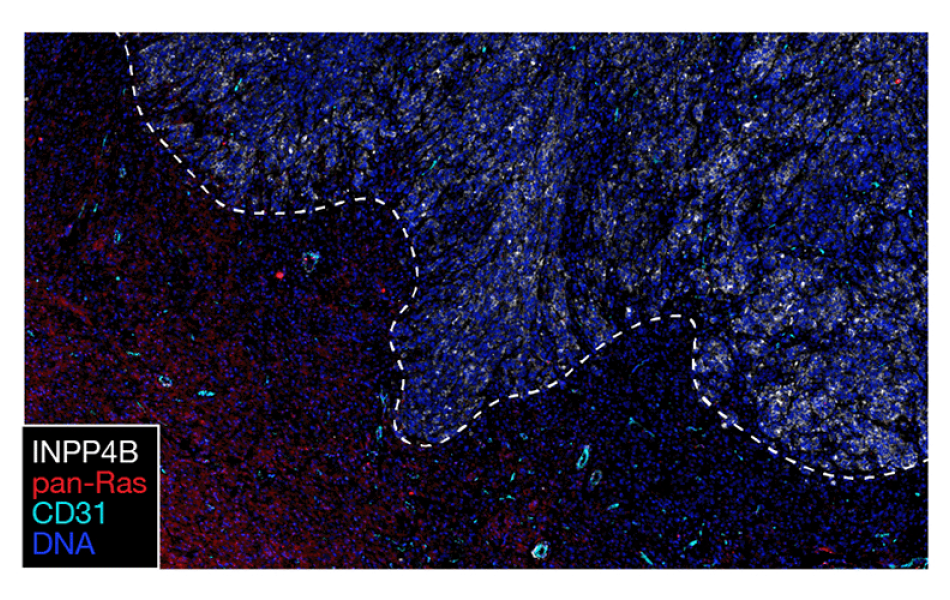
With new technology, Raleigh and his colleagues could visualize changes in protein expression and protein signaling events within the tumor sample. Image courtesy of the Raleigh lab.
One of the things that was really fun about this study was that it gave us the opportunity to use some really interesting and innovative technology on a really old problem,” he said. “It’s nice to be able to integrate all this information about the genomic architecture, single cell sequencing, and protein expression patterns in the context of the cells as they are found within the tumor.”
These newer spatial approaches for looking at meningiomas helped the researchers learn about the variation within the tumors and their response to therapy in unprecedented detail.
For example, by analyzing paired tissue samples from meningioma patients at initially diagnosis and at recurrence, the scientists found that new genetic mutations weren’t necessarily what was causing the tumor to grow back. Instead, the tumor cells turn on a series of cellular signaling pathways, and the new technology allowed the researchers to identify conserved themes among the newly activated genes and proteins.
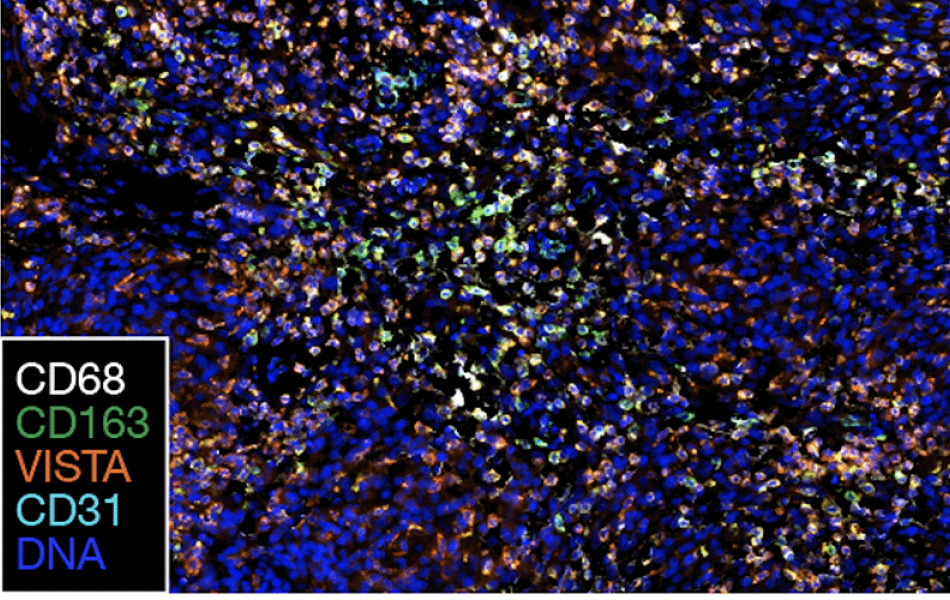
Spatial approaches helped the scientists identify conserved themes among the newly activated genes and proteins in newly diagnosed versus recurrent meningiomas. Image courtesy of the Raleigh lab.
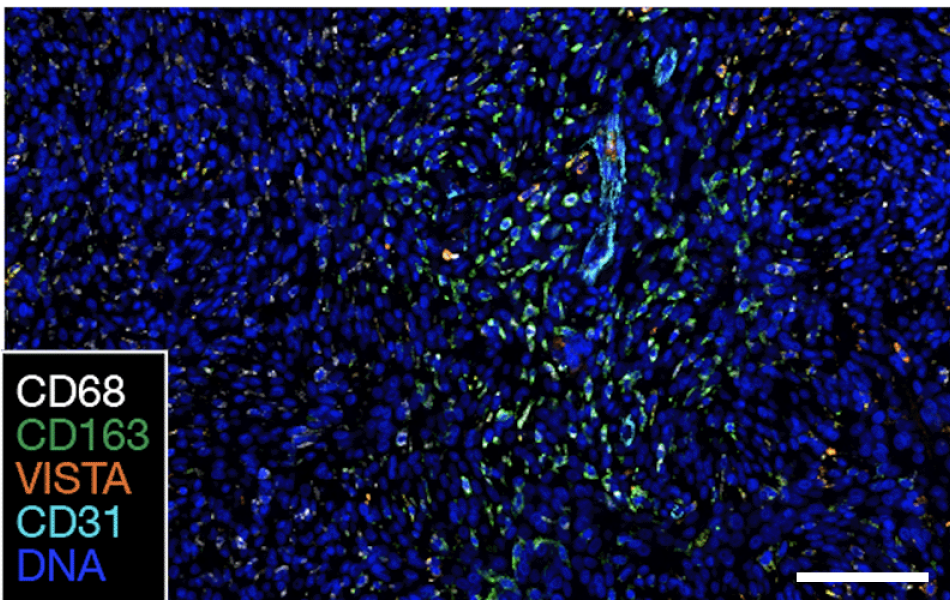
High-grade meningiomas have decreased immune infiltration at recurrence, as judged by the presence of the immune signaling markers CD45, VISTA and CD14. Image courtesy of Raleigh lab.
The scientists also confirmed their hypothesis that high-grade meningiomas with different driver mutations have different gene and protein expression patterns. These results suggest the current WHO classification scheme may need to be updated to stratify patients more accurately for clinical trials.
“We treat all these tumors the same way in currently available clinical trials, but that’s probably not the way we want to do these things moving forward,” Raleigh said.
Modeling meningiomas to test candidate treatments
The scientists then wanted to apply this newfound knowledge about high-grade meningiomas to test possible strategies to overcome treatment resistance.
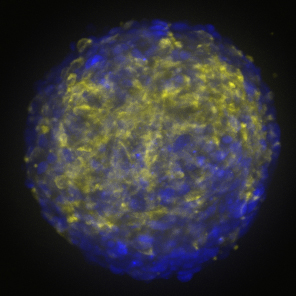
“Studying individual patients’ tumors is obviously really, really important, but that work will only get us so far without models to test our hypotheses about the therapies that we want to bring back to the clinic,” said Raleigh.
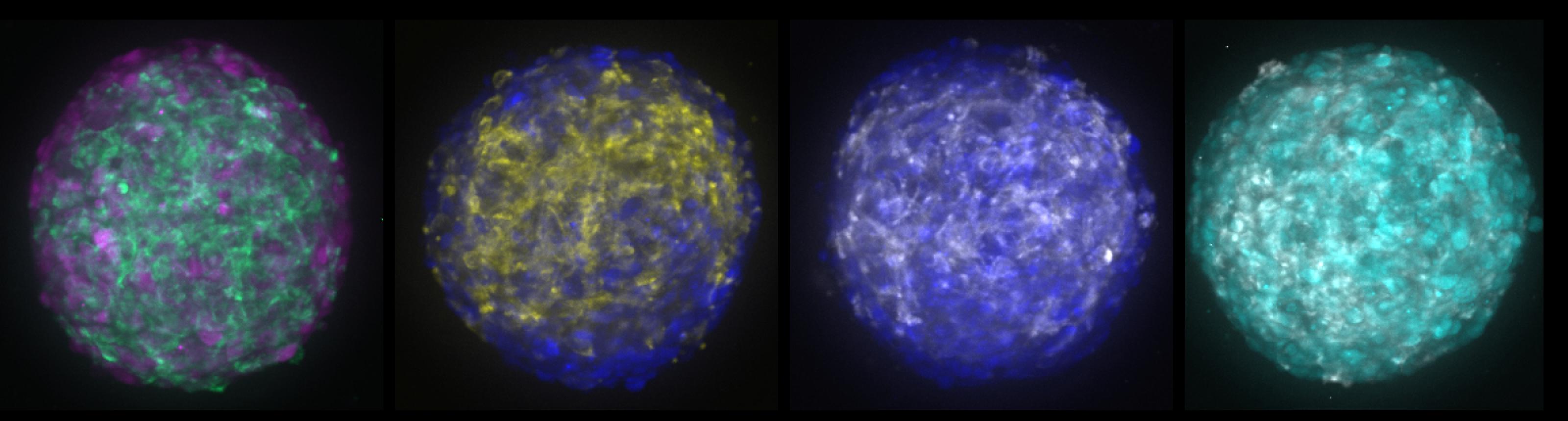
The scientists used a method called CRISPR interference to turn off different genes in meningioma cells grown in the lab. Raleigh’s lab modeled the tumors to represent different molecular groups of meningiomas they had previously identified. Each tumor cell population also had its own fluorescent marker so that the researchers could easily track how the different cells within the organoid were responding to various drugs.
“We now have these ‘meninge-oids’ with genetically predefined and genetically different cellular components that can serve as a platform for pharmacologic screening,” Raleigh said.
The most exciting results, he says, were when drug combinations specifically targeting each tumor cell population worked in tandem to block or even reverse the growth of the meningioma organoids.
He and his colleagues are now interested in using this system to develop models for each individual patient’s tumor. “It would allow us to pick the exact right combination of therapies,” he said.
Reference: Lucas, CH.G., Mirchia, K., Seo, K. et al. Spatial genomic, biochemical and cellular mechanisms underlying meningioma heterogeneity and evolution. Nat Genet (2024). https://doi.org/10.1038/s41588-024-01747-1