
New UCSF Study Identifies Precision Medicine Therapy for Rare Pediatric Glioma
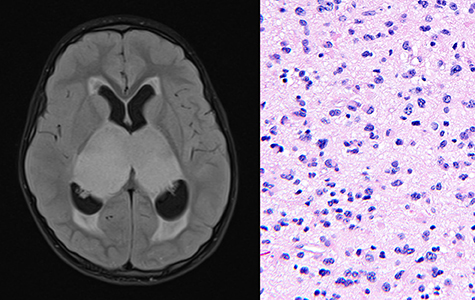
Brain tumors are the leading cause of cancer mortality in children, several types of which still do not have an optimal therapeutic strategy. Among these are diffuse gliomas, which cannot be cured by surgical resection alone due to the way the tumor cells infiltrate throughout the neighboring brain tissue.
Bithalamic gliomas – which involve both the right and left thalamus, a midline structure of the brain – are an unresectable and uniformly lethal diffuse glioma of childhood. “Very little was known prior to our study, and no effective treatments have been described to date,” said UCSF neuropathologist and cancer researcher David Solomon, MD, PhD.
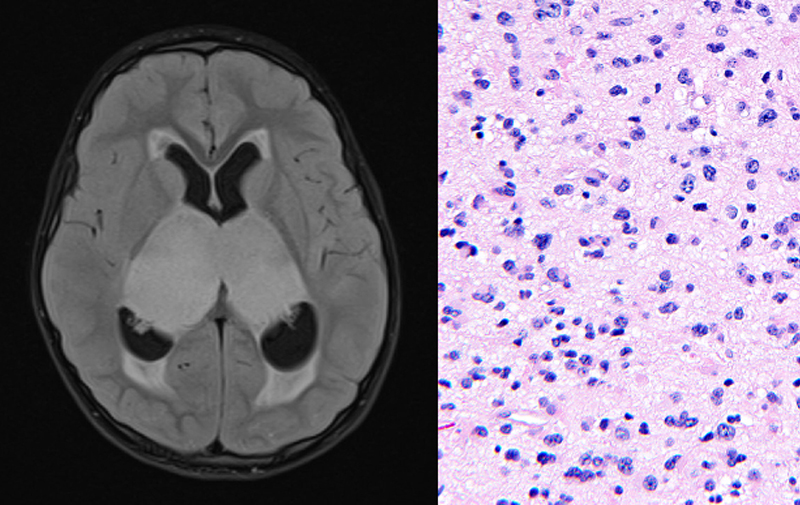
Published last week in the journal Acta Neuropathologica, Solomon and colleagues share a comprehensive genomic and epigenomic analysis of this rare tumor type, identifying gene alterations and a unique epigenetic signature that distinguish bithalamic glioma as a distinct tumor entity with potential sensitivity to a genomically-guided treatment strategy.
Genome-wide DNA methylation profiling of bithalamic glioma samples from 10 children was performed in the new UCSF Pathology Epigenomics Research Laboratory that is led by Solomon with support from the Departments of Pathology and Neurological Surgery, as well as the School of Medicine. This epigenetic analysis showed that bithalamic gliomas harbor DNA methylation patterns that are distinct from all glioma and other brain tumor types studied to date.
Additionally, targeted next-generation sequencing of bithalamic glioma samples from 13 children was performed at the UCSF Clinical Cancer Genomics Laboratory. This revealed that bilateral gliomas lack the histone H3 mutations that define midline diffuse gliomas arising in the unilateral thalamus (only on one side). Instead, Solomon and colleagues found that bithalamic gliomas are characterized by frequent mutations in exon 20 of the EGFR oncogene – which have not been found in any other gliomas to date.
Given the prevalence of EGFR mutations in bithalamic gliomas, Solomon and colleagues decided to investigate whether kinase inhibitors targeting EGFR would effectively block tumor growth. Using cultured human astrocytes engineered to express mutant isoforms of EGFR, they screened seven small molecule drugs that inhibit EGFR or MEK, a protein downstream of the EGFR signaling cascade. While all the inhibitors reduced survival of the mutant astrocyte cells, they discovered that efficacy of the individual drugs varied depending on the location and nature of the EGFR mutation.
Together, these data support an individualized strategy for kinase inhibitor treatment that is tailored to a patient’s specific EGFR mutation. Based on this rationale, four children with EGFR-mutant bithalamic gliomas were treated with a small molecule EGFR or MEK inhibitor as part of their therapeutic regimen. Those children whose treatment included a kinase inhibitor had a longer median survival of 21.5 months, compared to 13 months median survival for patients without kinase inhibitor treatment.
Although only a few children were part of this initial study, the results suggest a potential survival benefit with kinase inhibitor treatment, and further, establish the foundation for treating bithalamic gliomas using a precision medicine approach. Solomon is now working with colleagues at the Pacific Pediatric Neuro-Oncology Consortium (PNOC) to develop a randomized clinical trial for children with bithalamic gliomas to further test the efficacy of targeted kinase inhibition.
“This is one of a few select studies that I am the most proud of during my career here at UCSF,” said Solomon, who has spent the last eight years here, first as a resident, then fellow, and now faculty member. “It’s a multi-institutional, multi-disciplinary study that goes from patient to lab bench and then back to patient in a powerful way,” said Solomon.
Collaborators in this translational research study included physicians and researchers at St. Jude Children’s Research Hospital, Valley Children’s Hospital, Children’s of Alabama, University of California at Davis, Utah Primary Children’s Hospital, Nicklaus Children’s Hospital, and Children’s Hospital of Pittsburgh. This study was supported in part by the National Institutes of Health through the NIH Director’s Early Independence Award to David Solomon.
Reference
Mondal et al. (2020) Pediatric bithalamic gliomas have a distinct epigenetic signature and frequent EGFR exon 20 insertions resulting in potential sensitivity to targeted kinase inhibition. Acta Neuropathol. [Epub ahead of print]