
Newly identified cells may help support brain tumor microenvironment
UCSF Brain Tumor Center study indicates potential target for novel therapies
Glioblastomas (GBMs) develop resistance to current therapies thanks in part to the support of the many cells that make up the brain tumor microenvironment.
Scientists at UC San Francisco have now discovered a population of cells – known as cancer-associated fibroblasts – in the GBM microenvironment that may play an important role in helping these tumors grow.
Their findings, published March 1 in The Journal of Clinical Investigation, run counter to the scientific consensus that these cells are not found in brain tumors, presenting an unexplored avenue for new treatments.
“These would most likely be cells that could be targeted safely because they don't overlap with normal tissue,” said Manish Aghi, MD, PhD, a professor in the Department of Neurological Surgery and the study’s senior author.
Although cancer-associated fibroblasts are well-known to help support tumor growth in other cancers, scientists assumed that these cells did not exist in brain tumors.
“They were never really considered to be present in GBM since there are no normal fibroblasts in the brain,” Aghi said.
He added that many proteins in cancer-associated fibroblasts would also be present in the brain tumor cells – making the task of distinguishing them even more difficult.
Aghi and his colleagues used an established method for purifying cancer-associated fibroblasts from breast cancers, applying the protocol to tissue samples from glioblastoma patients. The researchers, led by Saket Jain, PhD, and Jonathan Rick, MD, found that the cells they had isolated from the tumors looked like cancer-associated fibroblasts under the microscope. These cells also did not have any of the chromosomal alternations that would be expected in tumor cells.
To verify that the cells were indeed cancer-associated fibroblasts, the scientists also performed single cell sequencing to analyze the abundance of all their genes. They calculated the probability that individual cells were cancer-associated fibroblasts based on the presence of nine genes typically used to identify them in other cancers and the absence of five genes common in lookalikes.
All these measures, Aghi said, demonstrated that between 50 to 60 percent of the cells they had isolated and cultured in petri dishes were cancer-associated fibroblasts.
The researchers then showed that these cells secreted factors that had strong stimulatory effects on GBM stem cells, which are known to influence brain tumor initiation and resistance.
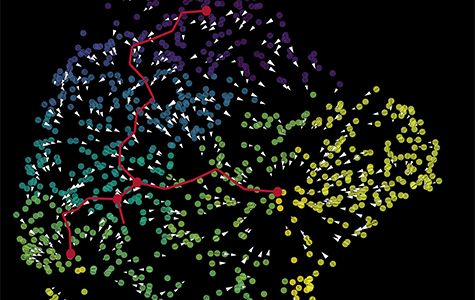
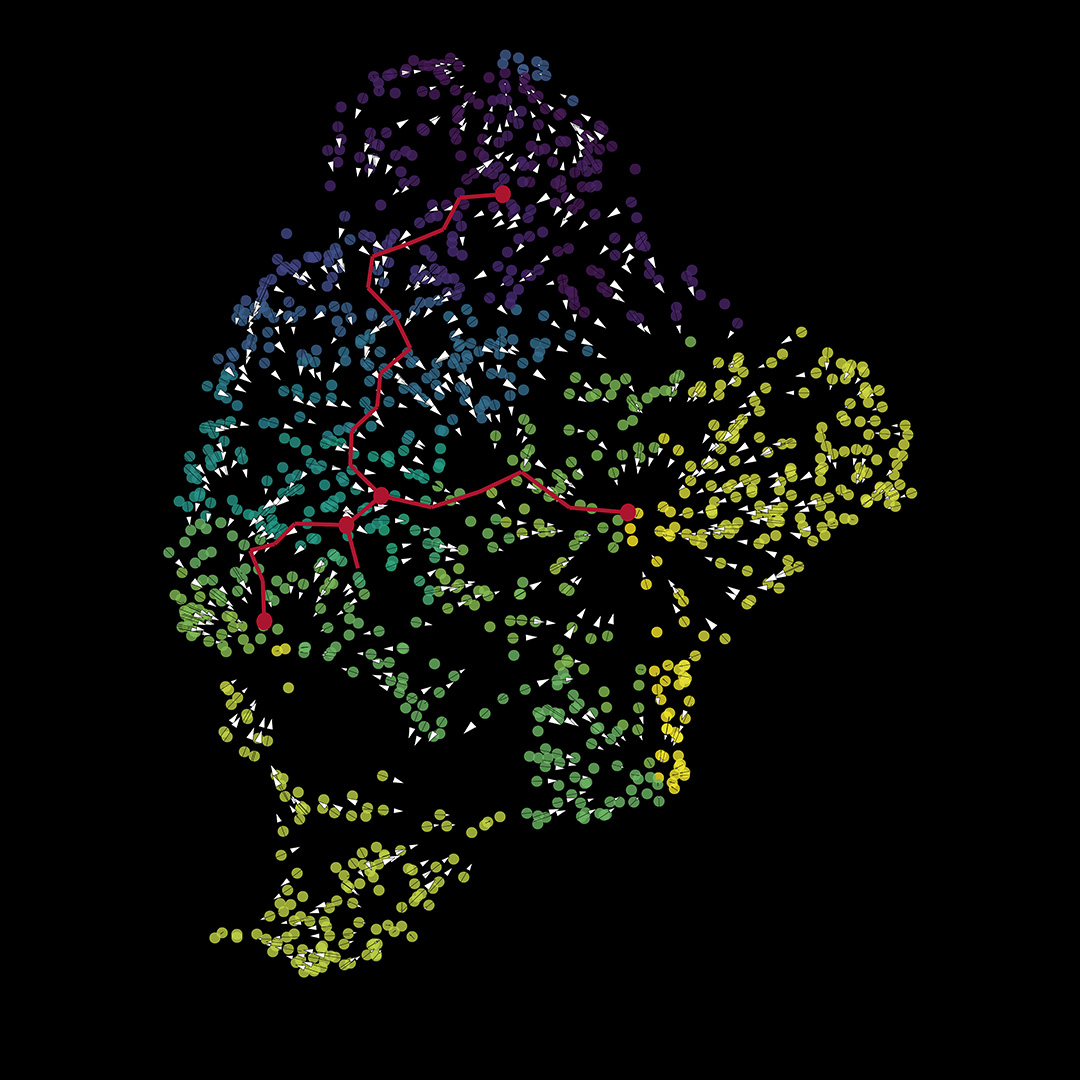
With a technique called spatial transcriptomics, the scientists could also see where within the tumors the cells they labelled as cancer-associated fibroblasts are located. Their analysis reveals that the cells are near both GBM stem cells and immune cells called M2 macrophages.
These results, according to Aghi, mean the cancer-associated fibroblasts are likely close enough to be exerting pro-tumoral effects on their neighboring cells.
Aghi and his colleagues are now looking to better isolate the cancer-associated fibroblasts from brain tumors by developing antibodies against proteins on the cells’ surface. With a pure population of cells, the researchers could further investigate how they work in GBMs. For example, he wants to figure out how much of the tumor mass they comprise and even screen for drugs that block their effects on the tumor.
For Aghi, whose lab has also examined neutrophils and natural killer cells, this research continues his group’s work in characterizing the understudied cells of the tumor microenvironment.
“It's a really exciting field to study because there are so many characters on the stage, each doing their own unique thing,” he said, “and each cell type to me represents a potential therapeutic opportunity.”
Paper(s) cited:
Jain S, Rick JW et al. Single-cell RNA sequencing and spatial transcriptomics reveal cancer-associated fibroblasts in glioblastoma with protumoral effects. J Clin Invest. 2023;133(5):e147087. doi.org/10.1172/JCI147087