
Immense Variation Found Within Meningiomas Offers Insight Into Therapeutic Targets
With 34,210 new diagnoses projected this year in the United States,1 meningiomas are the most common primary brain tumor. While the majority of meningiomas are low grade and typically respond well to surgery and radiotherapy, high grade meningiomas are still poorly understood and often recur despite treatment. All chemotherapy trials to date have failed to show benefit for these tumors, so new treatment targets are urgently needed.
“Differences within tumors called intratumoral heterogeneity has increasingly been recognized as a source of resistance to cancer treatments,” said UCSF physician-scientist David Raleigh, MD, PhD (Assistant Professor, Departments of Radiation Oncology and Neurological Surgery). For instance, different regions of an individual’s tumor can harbor different gene mutations, leading to different characteristics, behaviors, and treatment responses. Thus, a therapy that targets a specific mutation may only be effective for a subset of the tumor cells, allowing the tumor to continue growing despite targeting that specific mutation.
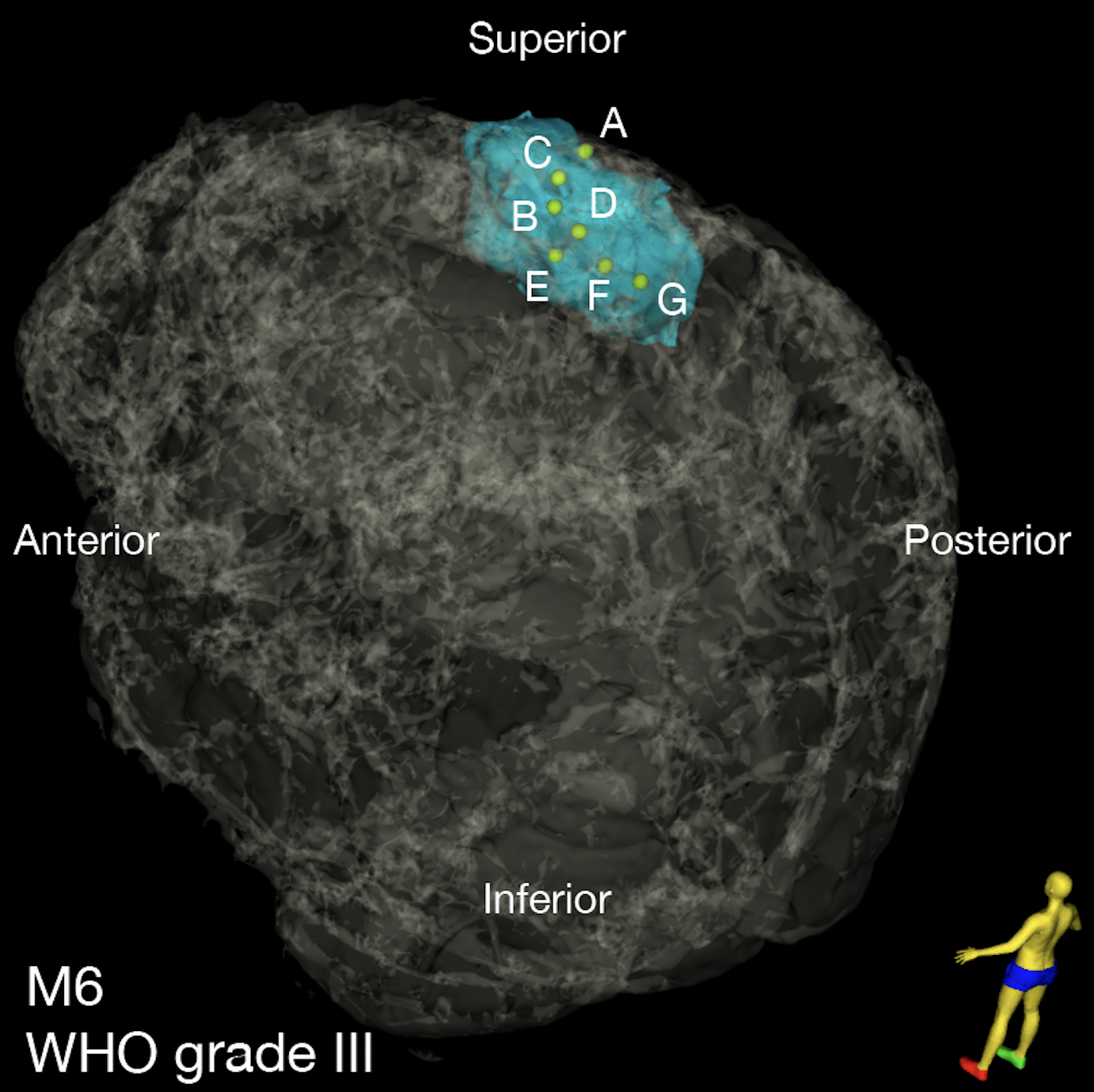
To better understand the extent of variation, or intratumoral heterogeneity, in meningioma, Dr. Raleigh and his team launched a comprehensive analysis of 86 meningioma samples, collected from separate locations within 13 patients’ tumors.
Published today in Nature Communications,2 Raleigh and colleagues leverage bioinformatics, histopathology, radiology, and even human cerebral organoids, to characterize the range of intratumor heterogeneity found in meningioma.
“We found that high grade tumors have greater heterogeneity than low grade tumors, which may explain their lack of response to existing treatments.” said first author and neurosurgeon Stephen Magill, MD, PhD, who recently graduated from the UCSF Neurological Surgery Residency Program. Dr. Magill is currently completing a minimally invasive cranial and endoscopic skull base fellowship at The Ohio State University.

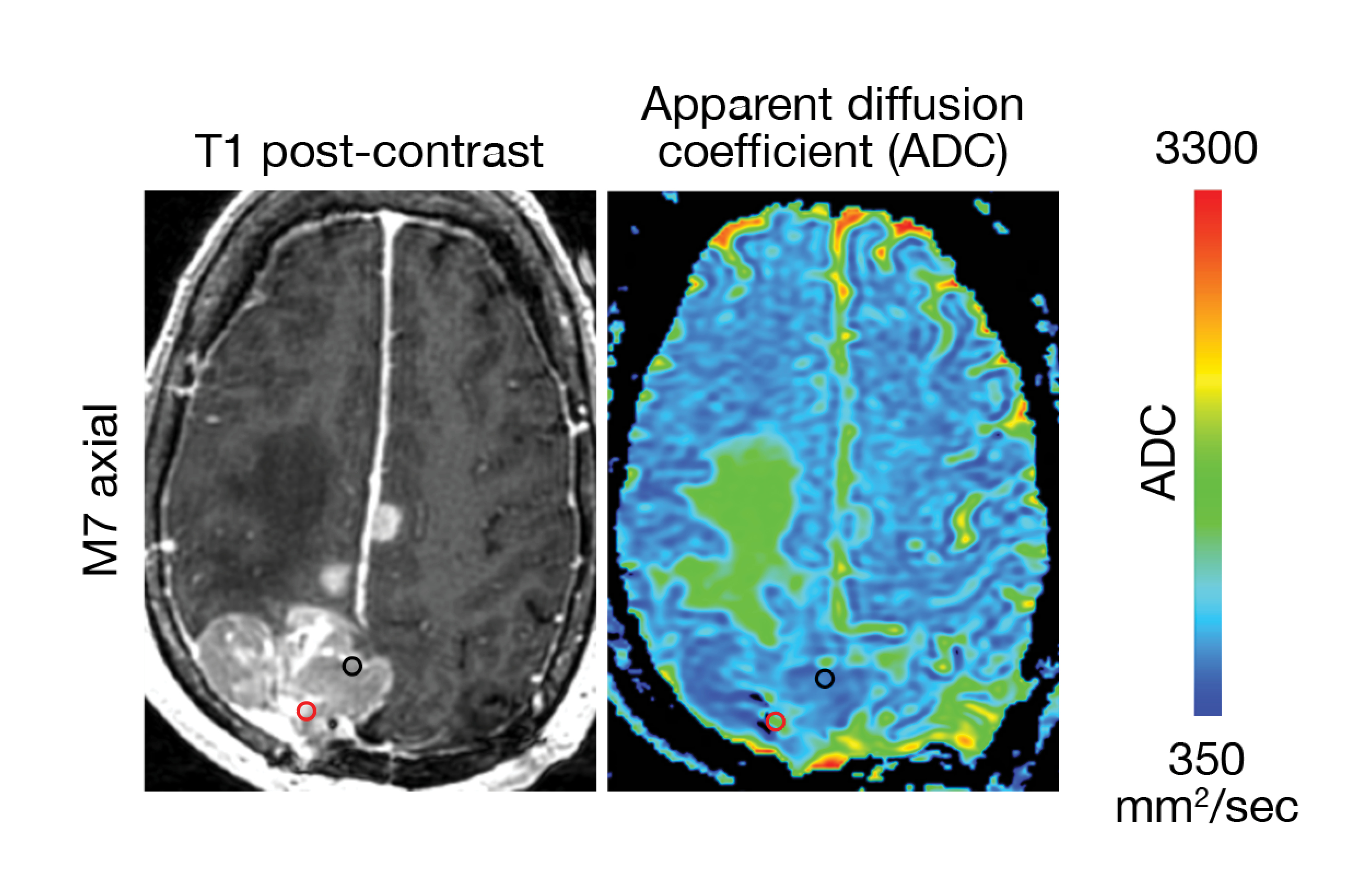
Magill and Raleigh further found that intratumoral heterogeneity in high grade meningiomas is associated with unique radiographic features, such as apparent diffusion coefficient (ADC), which is measured via MRI. Interestingly, they observed that regions of high ADC coincided with regions of proliferating tumor cells – and high expression levels of CDH2 and PTPRZ1, genes that are important for early brain development.
Next, Magill and colleagues demonstrate that genetic loss of CDH2 and PTPRZ1, through CRISPR interference, reduces proliferation of meningioma cells in culture. Similarly, exposure to ADH1, a small molecule inhibitor of CDH2, also inhibits proliferation of cultured meningioma cells.
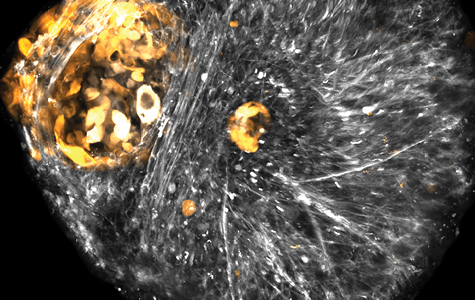
To further assess the role of CDH2 and PTPRZ1 in a more physiologically relevant context, the Raleigh Lab implemented a novel human cerebral organoid model of meningioma that was developed in collaboration with Eric Ullian, PhD and Daniel Lim, MD, PhD. Organoids are 3D cell cultures of brain tissue grown from human stem cells, and allow investigation of how tumor cells interact within the tumor microenvironment. They found that when low grade meningioma cells are grown with brain organoids, the meningioma cells form small spheres of cells next to the brain organoid, much like meningiomas grow around the surface of the brain. However, high grade meningioma cells invade into the brain organoid, much like high grade meningioma tumors.
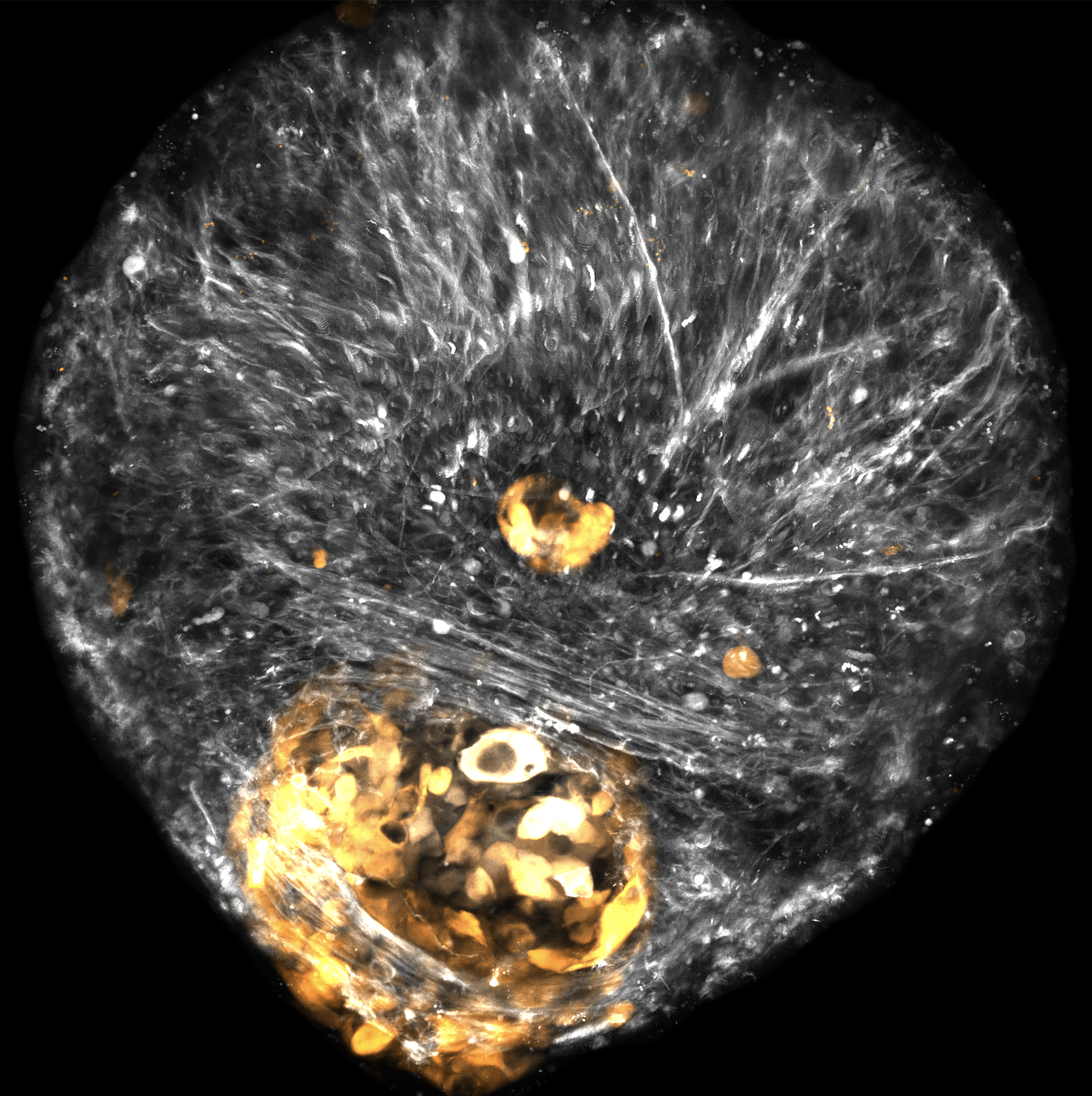
Raleigh and Magill found that meningioma cells not only express CDH2 and PTPRZ1 as they begin to surround and invade the cerebral organoids, but also that exposure to the inhibitor drug ADH1 blocks tumor formation.
Taken together, these findings identify CDH2 and PTPRZ1 as molecular drivers of meningioma intratumor heterogeneity, proliferation, and tumorigenesis that could be exploited as potential therapeutic targets. Moreover, the heterogeneous expression of CDH2 and PTPRZ1 – even within a single tumor – could provide a basis for why meningiomas grow asymmetrically and respond variably to treatment.
Looking ahead, Raleigh and Magill discuss how their work impacts discovery of future therapeutic targets, and carries potential implications for the clinical management and treatment of meningioma patients today.
“Given the significant intratumoral heterogeneity observed in high grade meningioma, image-guided sampling, particularly sampling and evaluating areas of high ADC signal, will be important for neurosurgeons and pathologists to predict how aggressive a tumor might be,” said Magill. “Work like this requires collaboration across many disciplines. For example, this project could not have been done without collaboration with Dr. Harish Vasudevan, resident physician in the Department of Radiation Oncology, or the organoid experiments that were done by Kyounghee Seo. It really speaks to the truly unique environment at the UCSF Brain Tumor Center, where I was fortunate to work with such a talented team of clinicians and scientists.”
“Working together, we will continue studying how meningiomas grow and behave so that we can develop better treatments for our patients,” added Raleigh.
References
- Ostrom QT, Cioffi G, Gittleman H, et al. CBTRUS Statistical Report: Primary Brain and Other Central Nervous System Tumors Diagnosed in the United States in 2012-2016. Neuro-oncology. 2019;21(Suppl 5):v1-v100.
- Magill ST, Vasudevan HN, Seo K, Villanueva-Meyer JE, Choudhury A, Liu SJ, Pekmezci M, Findakly S, Hilz S, LaStella S, Demaree B, Braunstein SE, Oberheim Bush NA, Aghi MK, Theodosopoulos PV, Sneed PK, Abate AR, Berger MS, McDermott MW, Lim DA, Ullian EM, Costello JF, Raleigh DR. Multiplatform genomic profiling and magnetic resonance imaging identify mechanisms underlying intratumor heterogeneity in meningioma. Nat Commun. 2020; 11:4803.