New Clinical Trial Targets Mutations Commonly Found in Lower Grade Gliomas
Within the next few months, the UCSF Brain Tumor Center will begin recruiting for a multi-center clinical trial to evaluate AG-881, a small molecule drug, for treatment of residual or recurrent IDH-mutant grade II glioma.
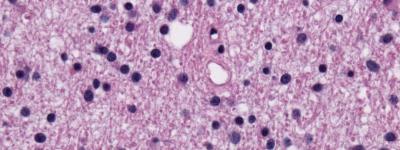
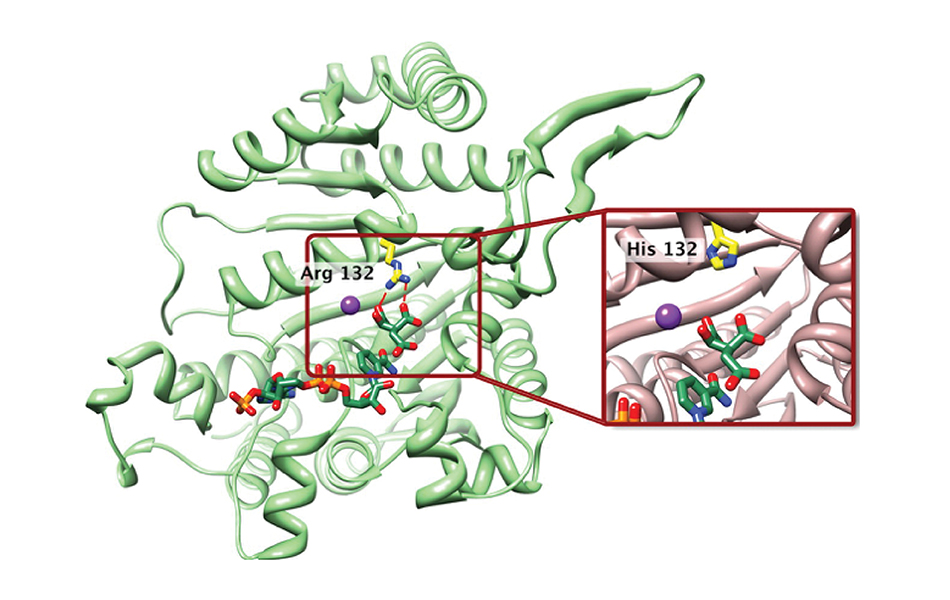
AG-881 is a drug that inhibits the mutated forms of the isocitrate dehydrogenase (IDH) 1 and 2 proteins. IDH1 and IDH2 gene mutations are commonly found across a variety of cancers, including glioma. In the last decade, the discovery of these mutations and their role in brain tumor growth and biology has led to a sweeping re-organization of the World Health Organization (WHO) classification system. Now, certain glioma subtypes are further defined based on their IDH mutation status – a distinction that greatly impacts tumor biology and patient outcomes.
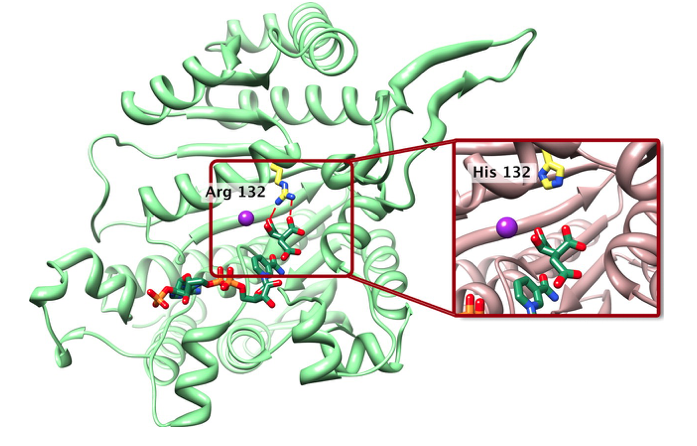
IDH mutations are known to be an early driver of glioma development, though the exact mechanism by which that occurs remains elusive. AG-881 acts to counter the overactive proteins produced within tumor cells carrying either of those mutations. “With IDH mutations present in the majority of low-grade gliomas, being able to specifically target this known driver of tumor development could have enormous impact,” says UCSF neuro-oncologist Jennifer Clarke, MD, who is leading the study at UCSF.

Preliminary findings from a phase I study of AG-881 show promising results for patients with IDH-mutant glioma. For patients with non-enhancing tumor, average six-month volumetric tumor growth was 6.8% following AG-881 treatment, compared to 24.5% in a similar glioma population without treatment. In addition, preliminary findings from a second, surgical study of AG-881 showed good brain penetration of the drug.
In this phase III trial, approximately 360 participants across multiple study sites will receive daily doses of the orally administered drug (or a placebo), with their treatment response followed. To be eligible, patients must have residual or recurrent grade II astrocytoma or oligodendroglioma with confirmed mutation(s) in IDH1 or IDH2, and be between 1 and 5 years from their most recent surgery.
For More Information on Enrollment
Study of AG-881 in Participants With Residual or Recurrent Grade 2 Glioma With an IDH1 or IDH2 Mutation
(415) 353-2966
Sign up to receive updates on clinical trials at the UCSF Brain Tumor Center.